Series
Igneous Rock Associations 10.
Komatiites
Jaroslav DostalDepartment of Geology Saint Mary’s University Halifax, NS, Canada, B3H 3C3
jdostal@smu.ca
Submitted March 2008. Accepted as revised, June 2008
SUMMARY
Komatiites are ultramafic volcanic rocks that occur mainly in Archean and Paleoproterozoic greenstone belts. These olivine-rich rocks are assumed to have crystallized from magmas that have about 28–30 wt% MgO. They are characterized by spinifex texture - platy or skeletal crystals of olivine set in a glassy matrix. Chemically, komatiites resemble peridotites and have high MgO but low SiO2, TiO2 (<1 wt%), K2O (<0.5 wt%) and incompatible trace element contents. Most of their compositional variations can be accounted for by olivine fractionation. Komatiites are conventionally considered to be derived from high temperature melts that have eruption temperatures of about 1600EC and are produced by high degrees of anhydrous melting of mantle plumes. The abundance of Archean komatiites, their decrease through the Proterozoic and extreme rarity in the Phanerozoic have been taken as evidence for secular cooling of the mantle. However, the plume model has recently been challenged. In marked contrast, it has been proposed that komatiites originate via hydrous melting at a shallow depth in subduction environments at significantly lower melting temperatures than those invoked by the plume hypothesis. This new model thus challenges traditional views of the early evolution of the Earth. Nevertheless, it appears that many komatiites are plume-related. In addition to the information they provide about the tectonics and the thermal evolution of Archean Earth, komatiites are economically important because they host locally significant magmatic Ni-sulfide (Ni-Cu-PGE) mineralization.SOMMAIRE
Les komatiites sont des roches volcaniques ultramafiques que l’on retrouve principalement dans des bandes de roches vertes archéennes et paléoprotérozoïques. On suppose que ces roches à fort contenu en olivine ont cristallisé à partir de magmas ayant des teneurs de 28 à 30% en poids de MgO. Typiquement, elles présentent une texture spinifex; c’est-à-dire en cristaux lamellaires ou squelettiques d’olivine dans une matrice vitreuse. Chimiquement, les komatiites ressemblent aux péridotites et présentent des teneurs élevées en MgO mais basses en SiO2, TiO2 (<1% en poids), K2O (<0.5% en poids) ainsi qu’en éléments traces incompatibles. La plupart de leurs variations compositionnelles peuvent s’expliquer par le fractionnement de l’olivine. Par convention, on considère que les komatiites proviennent de magmas de hautes températures qui ont des températures d’éruption d’environ 1600°C, et qui proviennent d0un haut degré De fusion de panaches mantéliques anhydres. L’abondance des komatiites archéennes, leur moindre abondance au Paléozoïque et leur extrême rareté au Phanérozoïque ont été interprétés comme étant l’effet d’un refroidissement séculaire du manteau. Par-centre, récemment, le modèle des panaches mantéliques a été remis en question. On a proposé qui au contraire les komatiites proviendraient de fusions hydratées de faibles profondeurs en milieux de subduction, et à des températures de fusion significativement plus basses que celles supposées par l’hypothèse des panaches. Ce nouveau modèle remet donc en question la perspective traditionnelle sur les premiers stades de l’évolution de la Terre. Ceci dit, il semble que nombre de komatiites soit relié à des panaches. Au-delà des informations qu’elles fournissent sur la tectonique et l’évolution thermique de la Terre archéenne, les komatiites sont importantes économiquement, étant les roches hôtes de minéralisations magmatiques de sulfures de nickel (Ni-Cu-EGP) en certains endroits.INTRODUCTION
1 Komatiites are rare ultramafic volcanic and subvolcanic rocks that occur, predominantly, in Archean and Paleoproterozoic greenstone belts. These rocks contain more than 18 wt% MgO (Le Bas 2000) and are distinguished from other magnesium-rich rocks, such as picrites and meimechites, by having spinifex texture (characterized by spectacular arrays of subparallel or randomly-oriented skeletal, platy and bladed olivine crystals set in a glassy groundmass; Fig. 1). Because all komatiites do not display spinifex texture, Arndt and Fowler (2004) defined komatiites as ultramafic rocks that either contain spinifex texture or are related to rocks that do (see Kerr and Arndt 2001). Komatiitic lavas are assumed to crystallize from magmas that have ultramafic compositions.
Figure 1. a) Komatiite flow showing a zone of spinifex-textured olivine blades overlying a cumulate zone (Pyke Hill, Munro Township, Ontario, Abitibi greenstone belt; photo courtesy of A. Fowler); b) Boulder of olivine spinifex-textured flow top komatiite. Top of flow is toward hammer handle. Olivine textures become rapidly coarser away from upper chilled margin of the flow and evolve from < 1 mm equant crystals (not visible) to radiate spinifex needles (2-10 mm long) to coarse bladed spinifex crystals (10-30 cm long; plate spinifex A3) in flow interior. Olivine textures are preserved by metamorphic mineral assemblage of serpentine + chlorite + tremolite (Spinifex stream area, Barberton Mountainland, Mpumulunga, South Africa; Dann and Grove 2007; photo courtesy of T. Grove). Hammer handle is ~ 0.5 m in length; c) Spinifex texture in thin section (Pyke Hill). Note skeletal spinifex-textured olivine crystals largely altered to serpentine with clinopyroxene spherulites (photo courtesy of A. Fowler).

Display large image of Figure 1
2 Komatiites were first identified by Viljoen and Viljoen (1969) in the Archean greenstone belt in the Barberton Mountainland of South Africa as a distinctive and "new" class of magnesium-rich (20–30 wt% MgO) volcanic rocks. Up until then, genuine ultramafic lavas were not known. Viljoen and Viljoen (1969) recognized that many high-Mg rocks in the Barberton greenstone belt were lava flows of significant lateral extent and thickness, with chilled or brecciated tops, amygdules, pillows and the distinctive quench textures that were subsequently named "spinifex" (after an Australian spiky grass - Triodia spinifex). The Viljoen brothers inferred that the rocks had characteristics of erupted liquids with distinctive chemical composition and named them after the Komati River flowing through the type locality. These 3.5 Ga flows are among the oldest known ultramafic rocks.
Figure 2. Proportions of komatiites in volcanic packages of greenstone belts as a function of age (modified after de Wit and Ashwal 1997, and Condie 2001).

Display large image of Figure 2
3 Similar ultramafic lavas were soon described from other Archean and Paleoproterozoic belts particularly from Canada, Africa, Australia and Finland. Most are Neoarchean to Paleoproterozoic (Fig. 2) but extend into the Phanerozoic, including Permian–Triassic komatiites from NW Vietnam (Glotov et al. 2001; Hanski et al. 2004) and Cretaceous (~ 90 Ma) komatiites from Gorgona Island off the coast of Columbia (Kerr et al. 1996; Kerr 2005; Brandon et al. 2003). However, komatiites have no known modern analogues. The predominance of komatiites in the Archean, their decreasing occurrence in the Proterozoic, and extreme rarity in the Phanerozoic have been interpreted to reflect secular cooling of the mantle (by up to 400EC; Nisbet et al. 1993). Although komatiites are considered to be important windows into Earth’s early mantle and therefore have major implications for constraining models of the thermal evolution of Earth, the temperatures, sources and depths of their formation remain controversial. In addition, komatiites are of economic interest as they are associated with significant magmatic nickel sulfide ore deposits in Australia and Canada.
GEOLOGICAL ENVIRONMENT
4 Although, where present, they occur only in minor/subordinate amounts, komatiites are an integral part of subaqueous volcanic successions of Precambrian greenstone belts. De Wit and Ashwal (1997) estimated that komatiites constitute typically less than 10% of the total volume of volcanic rocks in most greenstone belts although in some areas they form up to 30% of the volcanic succession (Fig. 2). Komatiite units can be tens or even hundreds of metres thick and can be traced continuously for up to 20 km, indicating that komatiite eruptions were major events during the Precambrian.
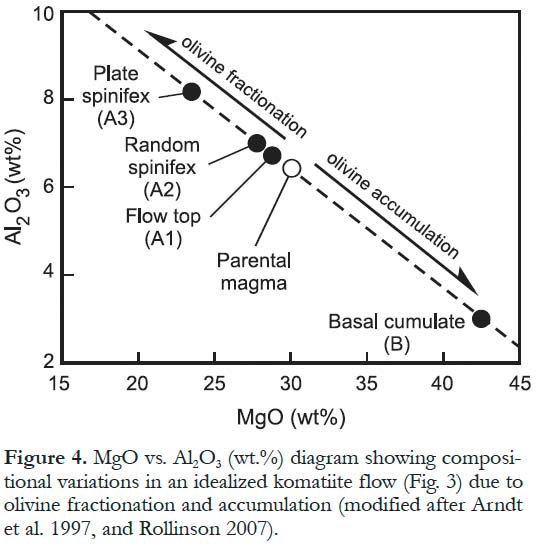
5 Komatiites occur either as lava flows or subvolcanic bodies; rarely, they are also pyroclastic. Komatiite lavas range from thin (a few cm) to massive to thick (> 100 m) and are distinctly layered. Individual flows displaying well-developed layering (Arndt et al. 2004; Grove et al. 1997) show a textural division that includes an upper part (Zone A), characterized by spinifex-textured rock, and a lower part (Zone B) containing a high proportion of equant olivine crystals resembling various peridotite to dunite cumulates (Fig. 3). Both zones have been further subdivided into several subzones, although the individual subzones may not be continuous and may occasionally be absent. Many thin and even some thick komatiite flows are not layered and do not have spinifex zones. They may have a massive texture, and olivine phenocrysts are commonly concentrated in the centre of such flows. Dann (2001) reported massive komatiite sheets, 12 to 50 m thick, traceable for more than 11 km and compositionally and texturally homogeneous. Massive lavas may pass laterally into flows with well-developed spinifex textures. The variation in the textures and layering of komatiite units has been attributed to variations in flow volume, lava flow velocity, effusion rate, topography and cooling rate (Sylvester et al. 1997; Dann 2001; Dann and Grove 2007), all of which might also be related to distance from an eruptive site.
Figure 3. Variations in texture across a several metre-thick komatiite flow showing well developed layering. The idealized section includes a thin (1-5 cm) chilled flow top (A1), a ~5–50 cm thick spinifex-textured subzone that has small randomly oriented spinifex blades usually millimetres to centimetres in length (A2 or random spinifex), and coarse-grained platy spinifex (A3) in which the olivine blades (centimetres to decimetres in length) are oriented perpendicular to flow margins and a cumulate zone (B). Zone B displays characteristics of crystal accumulation and inflation (ballooning). At the top of the Zone B, there may be a thin layer of foliated skeletal olivine (B1horizontal spinifex; probably rip-ups from A3). The rest of the Zone B includes a B2 subzone of massive medium- to fine-grained cumulate composed mainly of equant olivine crystals; B3 - coarse-grained equant olivine cumulate; B4 - massive cumulate composed of olivine crystals with chilled basal margin. Note that thicknesses of the zones may not be proportionally accurate.

Display large image of Figure 3
6 Komatiites are associated with komatiitic basalts (12–18 wt% MgO; Arndt et al. 1997), which have similar flow facies and textures as komatiites, except that the dominant phenocrysts are pyroxenes. Their differentiated flows contain upper pyroxene spinifex zones and lower cumulate zones.
7 Komatiite lavas are highly fluid because of their low viscosity and high liquidus temperature, so they would be expected to form rather thin flows. The thick komatiitic flows were probably ponded (lava lakes and rivers; Hill et al. 1995) or were thickened by flow inflation (flow ballooning) due to repeated flow pulses, like pahoehoe basalts of Kilauea, Hawaii (Dann 2001; Dann and Grove 2007). Komatiite lavas are inferred to have the ability to flow over considerable distances from eruption sites (Dann 2001; Sproule et al. 2002). Once a solid crust formed around a flow, it would create an efficient insulating surface that would facilitate the flow of hot komatiitic lava.
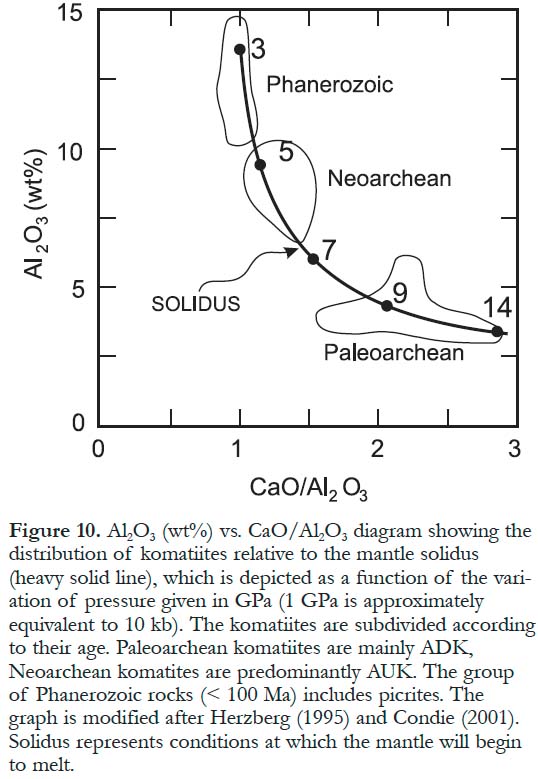
8 Some komatiitic rocks are intrusive; they usually form high-level dikes and sills and even layered differentiated bodies. These rocks have textures and compositions similar to those of their volcanic equivalents and might be the hypabyssal parts of the komatiitic volcanic suites (Arndt et al. 2004). Small amounts (typically <1%) of komatiitic volcaniclastic rocks have been documented in some greenstone belts (Sylvester et al. 1997).
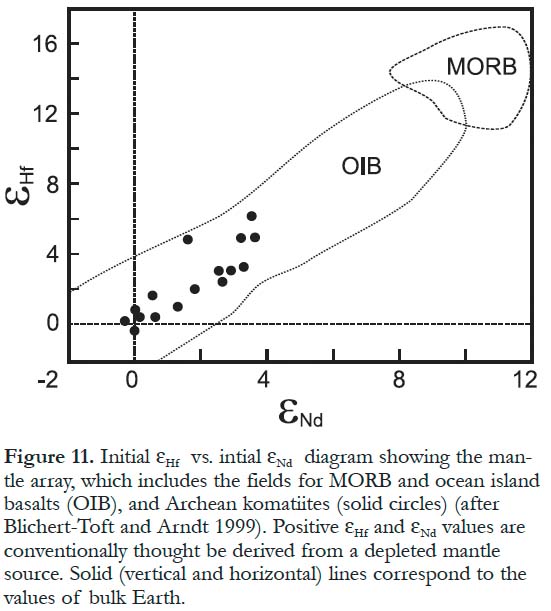
9 A variety of geological environments has been proposed for the emplacement of komatiites including mid-ocean ridges, plumes, oceanic plateaus, giant meteorite impacts and magma oceans (Grove and Parman 2004). There has been a suggestion that some komatiites are allochthonous, i.e. that they have been tectonically transported and interleaved with rocks of different environments (Sylvester et al. 1997). Most komatiites are associated with Archean and Paleoproterozoic greenstone belts (Arndt et al. 1997) where komatiites occur mainly in lithological assemblages that are considered to be remnants of ocean plateaus or arcs (Condie 2001). Some komatiites, including those from the quartzite–komatiite association that is widespread in the western Superior Province, overlie older granitic basement and are inferred to be autochthonous (Thurston and Chivers 1990; Bickle et al. 1994; Sylvester et al. 1997; Shimizu et al. 2004).
10 In Canada, some of the most prominent komatiitic occurrences are in the Abitibi belt, which contains several komatiitic successions that were emplaced over ca. 20 Ma (2724–2703 Ma). Komatiitic rocks of the Abitibi belt occur mainly in the Timmins– Kirkland Lake–Lake Abitibi region of Ontario and Québec.
MINERALOGY AND PETROGRAPHY
11 Komatiites and related rocks have been affected to variable degrees by metamorphism, hydrothermal and seafloor alteration, and deformation, which have, at least in part, obliterated the original textures and primary mineralogy. Hence, komatiites generally contain metamorphic minerals in place of their primary assemblages, relics of which may nonetheless be fortuitously preserved in some instances. Low-grade metamorphism of komatiites produced mineral assemblages dominated by serpentine–antigorite, chlorite, talc, tremolite, magnesite–dolomite and magnetite. At higher metamorphic grades, metamorphosed komatiites contain anthophyllite, enstatite, olivine and diopside.
12 The primary mineralogy of komatiites is simple: phenocrysts of olivine and spinel/chromite (+/pyroxene) are enclosed in a groundmass composed of glass, calcic clinopyroxene and minor orthopyroxene. In the spinifex-textured komatiites (Fig. 1), elongate and skeletal olivine (or pyroxene in komatiitic basalts) blades ranging in length from mm to tens of cm are set in a fine-grained matrix that originally contained a large proportion of volcanic glass ranging in composition from komatiite to komatiitic basalt (Donaldson 1982). Olivine is highly magnesian and shows normal zoning from cores of Fo95-90 to rims of Fo92-84. Their contents of Ni and Cr are high (up to > 4000 ppm and >2000 ppm, respectively; Donaldson 1982).
ORIGIN OF SPINIFEX TEXTURE
13 Petrologists who initially studied spinifex texture noticed that the skeletal olivine phenocrysts in komatiites (Fig. 1) resemble quench crystals (formed at very rapid cooling rates) in experimental melts (Donaldson 1982). Solidification under the conditions of supercooling (low nucleation rates and rapid crystal growth rates) produces a few large skeletal or dendritic crystals (Fig. 1). However, this model cannot explain the occurrence of spinifex texture within thick komatiite flows well below the upper chilled crust. Large skeletal crystals may have crystallized at depths, many metres below the surfaces of the flows (Fig. 3) where cooling rates must have been low. To overcome this problem, Viljoen and Viljoen (1969), among others, suggested that komatiites, most of which were related to submarine eruptions, were rapidly cooled by sea water. However, present day submarine basalts have only narrow rims that show features of rapid cooling and neither Archean basalts coeval with komatiites nor modern submarine basalts have spinifex textures. The origin of spinifex textures thus presents a dilemma: whereas the shape of crystals is suggestive of fast cooling near the flow margins, the blades formed deep within the flows.
14 As the spinifex texture is confined to MgO-rich basaltic to ultramafic rocks, a partial explanation is that spinifex texture is related to the temperature difference between the liquidus and solidus, which is very large for komatiites and Mg-rich rocks (400–500EC; Faure et al. 2006) compared with typical basaltic rocks (<100EC). However, in detail, the origin of the spinifex texture remains problematic. Because olivine crystals are thermally anisotropic, Shore and Fowler (1999) suggested that heat transfer increases the cooling rate in front of the crystal tips leading to the formation of platy crystals. Alternatively, a recent experimental study of Faure et al. (2006) inferred that spinifex texture is a result of slow cooling of ultramafic magma in a thermal gradient within a layer that separates magma from the solid outer crust. Another explanation has been put forward by Grove et al. (1997), who proposed that elevated water content in komatiitic magmas would lead to the rapid growth of large crystals, and accompanying degassing of hydrous komatiites would generate a strongly supercooled liquid. However, this hypothesis has been challenged (Arndt et al. 1998).
Figure 4. MgO vs. Al2O3 (wt.%) diagram showing compositional variations in an idealized komatiite flow (Fig. 3) due to olivine fractionation and accumulation (modified after Arndt et al. 1997, and Rollinson 2007).GEOCHEMISTRY
15 Virtually all komatiites and associated rocks have been metamorphosed and chemically altered. Many elements used in discussion of modern volcanic rocks such as Na, K, Rb, Sr and Ba as well as H2O and CO2 have commonly been redistributed. Thus geochemical/ petrological investigations of komatiites have been based upon elements that are considered to be less mobile, including Al, Ti, high-field-strength elements (HFSE) and rare-earth elements (REE).
16 Another potential complication is crustal contamination. Komatiitic magmas are prone to contamination because of their high temperatures. Crustal contamination of komatiite enriches light REE (LREE) and Th relative to heavy REE (HREE) and HFSE, particularly Nb and Ta (Jochum et al. 1991). Because of a significant compositional contrast between continental crust and komatiites, element ratios such as Nb/Th, Nb/U, Th/La and Nb/LREE are sensitive indicators of contamination (Jochum et al. 1991). Similarly, radiogenic isotopes, particularly Nd, provide useful constraints on contamination by older continental crust. The geochemical data suggest that crustal contamination did not play a major role in the genesis of most komatiites (e.g. Sproule et al. 2002).
Figure 5. Variations of MgO vs. Al2O3, TiO2, La, Gd, Th and Nb in Al-depleted (ADK) and Al-undepleted (AUK) komatiites (modified after Dostal and Mueller 2004). The solid and dashed lines delineate the ranges for AUK and ADK, respectively. AUK includes Abitibi, Belingwe and Kambalda greenstone belts while ADK encompasses samples from the Barberton and Abitibi belts.

Display large image of Figure 5
17 Komatiites have a chemical composition similar to peridotite or dunite, with high MgO, Ni and Cr but low contents of SiO2, TiO2 (<1 wt%), K2O (< 0.5 wt%), Na2O and incompatible trace elements. Arndt et al. (1997, 2004) inferred that original komatiitic liquids (i.e. lavas without olivine phenocrysts) had about 28–30 wt % MgO. This is close to the composition of aphyric komatiites and chilled margins of komatiitic flows. Olivine that crystallized from a parental magma of 28–30 wt% MgO would have a composition of ~ Fo94, similar to olivines in the lava flows (Arndt et al. 2004; Lesher and Arndt 1995).
18 Many komatiites show significant compositional variations, even within a single lava flow (Fig. 4). Most of these variations can be accounted for by fractionation of the first crystallizing mineral, olivine, involving either the gain or the loss of olivine phenocrysts. In differentiated flows, olivine cumulates at the base of the flows have 30–45 wt% MgO (Figs. 4, 5) whereas the spinifex-textured komatiites of the upper zone (residual liquids) have lower MgO (Fig. 4). This decrease of MgO in the differentiated komatiite flows from the basal cumulates to the spinifex zone is accompanied by an increase in Al2O3, TiO2, CaO and incompatible trace elements because these components are excluded from olivine (Fig. 5). Crystallization of olivine does not modify the element ratios as these elements will be enriched in the residual liquid to the same degree. The chondrite- and mantle-normalized trace-element patterns of the related spinifex and cumulate rocks are parallel although the former have higher absolute concentrations (Figs. 6, 7). Pyroxene spinifex-textured komatiitic basalts have lower MgO and Ni and frequently compositionally grade into komatiites.
Figure 6. Chondrite-normalized REE abundances in ADK and AUK komatiites. a) AUK from Pyke Hill, Munro Township (Fan and Kerrich 1997) compared to N-type mid-oceanridge basalts (N-MORB; Sun and McDonough 1989); S - spinifex AUK (Samples P-4, P-5, P-6 and P-8), C - cumulate AUK (Samples P-2 and P-3); b) ADK from Komati Formation, Barberton greenstone belt (Lahaye et al. 1995). S - spinifex ADK (Samples 166-1, 229-2, 229-3, B12 and B14), C- cumulate ADK (samples 229-5 and B15). Normalizing values after Sun and McDonough (1989).

Display large image of Figure 6
19 According to their chemical composition, komatiites are usually subdivided into two major types (Nesbitt et al. 1979; Arndt et al. 1997): aluminum (Al)-undepleted (or Munrotype, named after their type locality in the Abitibi greenstone belt) and aluminum-depleted (or Barberton-type). Al-undepleted komatiites (AUK) are characterized by ratios of Al2O3/TiO2 ~ 20 and CaO/Al2O3 ~ 1, values that are similar to those of chondritic meteorites and primitive mantle. Their REE patterns (Fig. 6) are typically slightly depleted in LREE and have a flat HREE segment with (Gd/Yb)n ~ 1 (nchondrite-normalized). They resemble the patterns of recent N-type midocean ridge basalts (MORB) although the absolute REE abundances in komatiites are significantly lower than in MORB (Fig. 6). The mantle-normalized patterns of AUK are also characterized by near chondritic Ti/Zr (~110) and depletion of Th, Nb and LREE (Fig. 7). Al-undepleted komatiites are the most widespread komatiitic lavas and occur predominantly in 2.7 Ga Archean greenstone belts including the Abitibi (Ontario-Québec), Belingwe (Zimbabwe) and Norseman-Wiluna (Australia) but are rare in pre-3.0 Ga greenstone belts. They include the 2718–2710 Ma Kidd– Munro assemblage (eastern Ontario), which contains the ~1000 m-thick Munro komatiite flows. Some AUKs are also of Proterozoic and Phanerozoic ages such as those from Gorgona Island (Kerr et al. 1996).
20 Al-depleted komatiites (ADK) are lower in Al2O3 and have low Al2O3/TiO2 (typically <12; Fig. 8) but high CaO/Al2O3 (~2–2.5). Their REE patterns (Fig. 6) have fractionated HREE with (Gd/Yb)n >1.3. Al-depleted komatiites have higher contents of strongly incompatible trace elements (Th, LREE) than the AUK but their mantle-normalized patterns typically show small negative Zr and Hf anomalies (Fig. 7). Al-depleted komatiites are the most abundant komatiitic lavas in the 3.5–3.0 Ga Barberton greenstone belt of South Africa and the ~3.5 Ga greenstone belts of the Pilbara craton (Australia) but in other greenstone belts, particularly those of post-3.0 Ga age, they are rare. An association of both AUK and ADK is uncommon but has been documented in the Abitibi greenstone belt (e.g. Dostal and Mueller 1997; Fan and Kerrich 1997) among other places. Komatiitic basalts include both Al-undepleted and Aldepleted types and their characteristics match those of komatiites. In addition to the two komatiite groups, several other komatiite subtypes have been recognized in particular regions but they are only subordinate (e.g. Sproule et al. 2002).
Figure 7. Primitive mantle-normalized trace-element abundances in ADK and AUK komatiites. a) AUK from Alexo, Ontario (Lahaye and Arndt 1996) compared to N-type MORB (Sun and McDonough 1989). S - plate spinifex AUK (samples 656, 657, 665, 668), C - basal cumulates (samples 712, 714 and 715); b) ADK from Komati Formation, Barberton greenstone belt (Lahaye et al. 1995). S - spinifex ADK (samples 166-1, 229-2, 229-3, B12 and B14), C - cumulate ADK (samples 229-5 and B15). Normalizing values after Sun and McDonough (1989).

Display large image of Figure 7
21 The Nd isotopic data on relatively fresh komatiites as well as their fresh pyroxenes from the Abitibi belt yielded GNd values of 2.8 to 3.8 (e.g. Lahaye et al. 1995). Some komatiites have slightly higher GNd values (e.g. Lesher and Arndt 1995).
Figure 8. Element ratios distinguishing AUK and ADK. The data for the AUK and ADK fields were compiled from literature, mainly by Dostal and Mueller (2004). Solid lines represent chondritic (primitive mantle) values (n-chondrite-normalized; chondritic values after Sun and McDonough 1989).PETROGENESIS
Melting Conditions
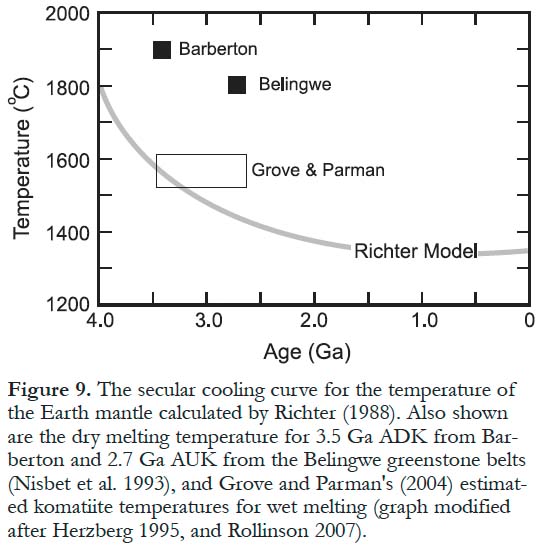
22 From the time of their discovery, there has been active debate about the melting conditions required to produce liquids of komatiitic composition. Because the MgO contents of anhydrous magmas are proportionally related to their melting temperature (higher MgO results from higher melting temperatures), komatiite melts require significantly higher temperatures (Green et al. 1975) than the 1250–1350EC responsible for recent MORB. Traditionally, most petrologists have accepted that during the Archean and Paleoproterozoic, komatiite melting temperatures ranged from 1600–1900EC, so komatiitic magmas were therefore related to deep-seated mantle plumes (Campbell et al. 1989). Plumes are hot upwelling jets in the mantle that originate as temperature instabilities at thermal boundary layers, and possibly may include material from the core–mantle boundary and transition zone. Alternatively, these high temperatures led some researchers to propose that the komatiitic melts were produced by hydrous melting of the mantle wedge above subduction zones, where the presence of water significantly decreased the melting temperature (Grove et al. 1997; Grove and Parman 2004). This model challenges the view that the Archean mantle was unusually hot (Fig. 9). The controversy regarding "wet" (subduction-related origin) versus "dry" (mantle plume origin) melting is still very much alive.
Figure 9. The secular cooling curve for the temperature of the Earth mantle calculated by Richter (1988). Also shown are the dry melting temperature for 3.5 Ga ADK from Barberton and 2.7 Ga AUK from the Belingwe greenstone belts (Nisbet et al. 1993), and Grove and Parman's (2004) estimated komatiite temperatures for wet melting (graph modified after Herzberg 1995, and Rollinson 2007).
Display large image of Figure 9
23 Currently, the conventional model among petrologists is that komatiites are high temperature, low viscosity melts produced by high degrees of anhydrous melting of mantle plumes. They erupted at temperatures close to 1600EC. Because CaO/Al2O3 is strongly pressure dependent, Herzberg (1995) used an Al2O3-CaO/Al2O3 plot to estimate the depth of melting of komatiite magmas. Figure 10 shows that the Paleoarchean komatiites (mainly ADK), which have low Al2O3 and high CaO/Al2O3, were generated at depths of 300–450 km (9–14 GPa), Neoarchean komatiites (mainly AUK) from depths of 150–200 km (5–6.5 GPa), and young komatiites (< 100 Ma old) from depths of 100–130 km (3–4 GPa). Nisbet et al. (1993) proposed similar although slightly higher pressure conditions; they inferred that komatiitic (AUK) magmas that have about 26wt % MgO would have an eruption temperature of 1520EC and have melted at a temperature of 1800EC at a depth of about 12 GPa (~370 km), whereas ADK with about 29 wt% MgO, had an eruption temperature of about 1580EC and melted at 1900EC at a depth of 18 GPa (~ 560 km; i.e. in the majorite garnet stability field; majorite garnet is a high pressure Mg-silicate mineral which occurs in the mantle transition zone, below 400 km).
24 Most Archean plumes are inferred to have been composed of depleted mantle material (Campbell et al. 1989; Lesher and Arndt 1995). The low concentrations of incompatible trace elements and their patterns (Fig. 7), and element ratios in both komatiitic types are, in general, consistent with their derivation from sources depleted in incompatible elements relative to a primitive mantle composition, i.e. depleted by a previous melt extraction event (low degree [1–2%] partial melts; Hofmann 1988). Likewise, Nd isotopic characteristics of komatiites point to derivation from a depleted mantle source (Fig. 11). The high contents of MgO (up to 30 wt%) coupled with low abundances of incompatible trace elements in komatiites suggest that the komatiitic magma was generated by a high degree of partial melting when Mg-rich olivine and orthopyroxene were dominant phases entering the melts. Herzberg (1992) inferred that the komatiites were formed by 30–50% partial melting of mantle peridotite.
Figure 10. Al2O3 (wt%) vs. CaO/Al2O3 diagram showing the distribution of komatiites relative to the mantle solidus (heavy solid line), which is depicted as a function of the variation of pressure given in GPa (1 GPa is approximately equivalent to 10 kb). The komatiites are subdivided according to their age. Paleoarchean komatiites are mainly ADK, Neoarchean komatites are predominantly AUK. The group of Phanerozoic rocks (< 100 Ma) includes picrites. The graph is modified after Herzberg (1995) and Condie (2001). Solidus represents conditions at which the mantle will begin to melt.
Display large image of Figure 10
25 An alternative model invokes wet mantle melting in subduction environments. Modern plume magmas typically contain only very small amounts of water (<0.5 wt%; Dixon et al. 2002) and modern hydrous melts are related to volatiles released from the subducting slabs. Grove and his colleagues (Grove and Parman 2004; Grove et al. 1997; Parman et al. 1997) argued that wet mantle melting can produce komatiitic melts at temperatures well below 1600EC as compared to 1800 and 1900EC advocated by Nisbet et al. (1993). Experimental studies (e.g. Inoue et al. 2000) have shown that hydrous melting can generate ADK at relatively low temperatures (1300–1500EC). The experiments of Barr and Grove (2006) suggest that the Barberton komatiites were formed by wet melting at depths lower than those corresponding to pressures of 1.7 GPa (< 55 km). This would also imply that the mantle during the Archean was not significantly hotter that at the present (Fig. 9), probably by only about 100ºC (Grove and Parman 2004).
Figure 11. Initial EHf vs. initial ENd diagram showing the mantle array, which includes the fields for MORB and ocean island basalts (OIB), and Archean komatiites (solid circles) (after Blichert-Toft and Arndt 1999). Positive EHf and ENd values are conventionally thought be derived from a depleted mantle source. Solid (vertical and horizontal) lines correspond to the values of bulk Earth.
Display large image of Figure 11
26 Grove and Parman (2004) argued that the presence of vesicles in komatiites and the occurrence of pyroclastic komatiites is evidence for the degassing of erupting hydrous komatiites, whereas Parman et al. (1997) concluded that high pre-eruptive water contents are needed to reproduce observed pyroxene compositions in the Barberton komatiites. They inferred that pyroxenes from Barberton komatiites crystallized from a magma having about 6 wt% water. The wet melting model is also supported by analyses of melt inclusions that have escaped metamorphism. Shimizu et al. (2001) documented the presence of water in komatiitic melts. Because some Archean komatiites are associated with calc-alkaline volcanic rocks and boninites, Grove and Parman (2004) argued that like recent boninites, komatiites may result from wet melting above a subduction zone. However, the wet melting model has been fiercely contested. Arndt et al. (1998) noted that low viscosity komatiite melts cannot retain dissolved volatiles. Also, a depletion of incompatible elements in most komatiites requires a depleted mantle source (i.e. a source which underwent previous melt extraction). As water behaves as an incompatible element during melting, it would be expected to have been consumed during the depletion event and would not be present during melting producing komatiite magma (Sproule et al. 2002). Bouquain et al. (2006) also disputed the pyroxene argument of Parman et al. (1997). Thus there is a continuing debate on the origin of komatiites.
27 Several processes have also been invoked to explain the relationship between komatiites and komatiitic basalts. Komatiitic basalts can be derived from komatiites by fractional crystallization or by fractional crystallization accompanied by crustal contamination. However, Sproule et al. (2002) argued that the relatively uniform compositions of komatiitic basalts in the Abitibi greenstone belt, accompanied by a lack of evidence of contamination, do not support an assimilation–fractional crystallization model. Alternatively, komatiitic basalts can be derived from a similar source as komatiites but by a lower degree of melting (Dostal and Mueller 1997).
Al-depleted vs. Al-undepleted Komatiites
28 Most recent models for the origin of komatiites postulate that differences between ADK and AUK are related to the role of garnet in their sources (e.g. Herzberg 1995; Arndt et al. 1997), although the sources of both komatiite types are isotopically (Lu-Hf and Nd-Sm) similar (Blichert-Toft and Arndt 1999). Al-undepleted komatiites, which have flat HREE patterns and chondritic values of Al2O3/TiO2 (Fig. 8) and Ti/Zr, did not have garnet in the melting residue. This implies that garnet, which can fractionate these elements, was either incorporated into the liquid during melting or was not present in the source. Al-undepleted komatiites are thought to be produced by a large amount of melting of a garnet peridotite leaving only olivine (+/-or orthopyroxene) in the residue (Arndt et al. 1997). Alternatively, AUK could have been generated by melting of a garnet-free source either at a shallow depth (above the garnet stability field; < 3 GPa) or at great depth, below the mantle transition zone (> 660 km) where perovskite (a major silicate mineral in the lower mantle) becomes the major mantle phase (Xie and Kerrich 1994).
29 Low Al2O3/TiO2 in ADK (Fig. 8), as well as the fractionated HREE patterns, indicate that either their mantle source was depleted in Al, or garnet remained in the residue after the melting and extraction of ADK. The latter is supported by Lu-Hf isotopic data (Blichert-Toft and Arndt 1999) which show that the Barberton ADK was derived from a garnet-bearing source and their residuum was garnet-rich. Thus ADK formed either in the presence of garnet at depths of 100–400 km or majorite garnet at depths of 400–660 km (possibly a plume tail; Campbell et al. 1989). Xie and Kerrich (1994) inferred that the trace-element signatures, particularly the ratios of HFSE (Zr, Hf, Nb) to the REE can distinguish between olivine (shallow mantle), majorite (>14 GPa; >~ 400 km; Herzberg 1995) and perovskite (lower mantle) fractionation. The negative Zr-Hf anomalies observed in ADK (Fig. 7) were interpreted to be due to melting with residual majorite garnet.
MINERALIZATION ASSOCIATED WITH KOMATIITES
30 Komatiites and komatiitic basalts locally host magmatic Ni-sulfide (Ni-Cu-PGE) mineralization. Famous deposits include those in the Archean Yilgarn and Pilbara cratons of Western Australia, the Abitibi greenstone belt, Zimbabwe craton, the Proterozoic (1.8 Ga) Cape Smith (Ungava) belt of Québec and the Proterozoic Thompson belt of Manitoba. Principal ore minerals are pyrrhotite, pentlandite, pyrite and chalcopyrite. The sulfides formed as immiscible liquids and thus they are of primary magmatic origin. The production of large amounts of immiscible sulfide droplets that settle in the host magma to form an orebody requires the attainment of sulfide saturation in the komatiitic melts, mainly through the assimilation of sulfur from sulfurrich country rocks. Nickel, copper and platinoid elements as well as iron would then be scavenged by the sulfur to form sulfide droplets.
31 Naldrett (2004) subdivided these komatiite-hosted deposits into three groups. The first type includes ore bodies that are generally small (1 to 5 x 106 tonnes) but high grade (1.5–3.5 wt% Ni) and occur where massive sulfides are concentrated at the base of the host ultramafic flows in zones up to about 50 m thick. Examples include the 2.7 Ga deposits of the Kambalda district (with 0.8–1.4 wt% Cu), which lie in the Yilgarn craton, and the Langmuir deposit of the Abitibi greenstone belt, about 40 km southeast of Timmins, Ontario.
32 The second deposit type encompasses large (100 to 250 x 106 tonnes) but low grade (~0.6 wt% Ni) deposits of disseminated sulfides in olivine-rich cumulate dikes/sills that feed komatiitic flows. Examples of these deposits are the Six Mile and Mt. Keith deposits near Yakabindi, Western Australia and the Dumont deposit of Québec (~ 60 km NE of Rouyn). The third type includes deposits related to Proterozoic komatiitic basalts that occur in rifted continental margin environments. They host deposits of the Thompson (Manitoba) area and the Cape Smith fold belt (Ungava Peninsula, Québec).
CONCLUSIONS
33 Komatiites provide a window into the composition and thermal dynamics of the Archean mantle. They are usually considered to be the product of mantle plumes generated at depth, possibly near the core–mantle boundary (Campbell et al. 1989; Arndt et al. 1998; Sproule et al. 2002). The chemical compositions of komatiites and related Mg-rich rocks appear to vary with time. For example, with respect to the MgO contents of common ultramafic magmas, komatiites characteristic of the Archean have >18 wt% MgO, komatiitic basalts dominant in the Proterozoic have 12–18 wt% MgO and picrites in the Phanerozoic have 10–13% wt% MgO. Because the MgO contents of dry magmas are related to their melting temperature, the trend is consistent with secular cooling of the Earth’s interior. However, recently, some researchers (Parman et al. 1997; Grove and Parman 2004; Wilson et al. 2003) have challenged the plume model and attendant volatile-free mantle melting, and postulated that komatiites may be produced by hydrous melting at shallow mantle depth in a subduction (arc) environment. In this scenario, temperatures would be considerably lower than those required by the plume model. Since many current models of chemical differentiation and thermal evolution of the Earth are based on mantle plume-generated komatiites, the subduction model, if correct, would significantly change current views of the evolution of Archean Earth. Although recent data imply that many komatiites are derived from a dry mantle source, there are still some rocks that could be subduction-related, such as high-SiO2 komatiites from the ~3.33 Ga Commondale greenstone belt in South Africa (Wilson et al. 2003), and some komatiitic basalts that resemble boninites. More studies of the trace element and isotopic characteristics of komatiites and their minerals as well as experimental studies, particularly under wet melting conditions, are needed to constrain their origin. Obviously, there is much left to learn about these fascinating rocks.
ACKNOWLEDGEMENTS
I am grateful to Victor Owen for reading an early draft of the manuscript, to Andrew Hynes and Tony Fowler for thorough, constructive reviews and to Georgia Pe-Piper for editorial advice and continuous encouragement. I have benefited greatly from discussions with Wulf Mueller. Many thanks go to Tim Grove, Tony Fowler and Michel Houle for the photos of komatiites and Randy Corney for cheerful technical assistance. The continuing support of the Natural Sciences and Engineering Research Council of Canada is gratefully acknowledged.REFERENCES
Arndt N.T., and Fowler, A.D., 2004, Textures in komatiites and variolitic basalts, in Eriksson, P.G., Altermann, W., Nelson, D.R., Mueller, W.U. and Catuneanu, O., eds., The Precambrian Earth: Tempos and Events: Developments in Precambrian Geology, v.12, Elsevier, Amsterdam, p. 298-311.
Arndt, N.T., Albarede, F., and Nisbet, E.G., 1997, Mafic and ultramafic magmatism, in de Wit, M.J. and Ashwal, L. D., eds., Greenstone Belts: Oxford University Press, Oxford, p. 233-254.
Arndt, N.T., Ginibre, C., Chauvel, C., Albarede, F., Cheadle, M., Herzberg, C., Jenner, G., and Lahaye, Y., 1998, Were komatiites wet?: Geology, v. 26, p. 739-742.
Arndt, N.T., Lesher, C.M., Houle, M.G., Lewin, E., and Lacaze, Y., 2004, Intrusion and crystallization of a spinifextextured komatiite sill in Dundonald Township, Ontario: Journal of Petrology, v. 45, p. 2555-2571.
Barr, J.A., and Grove, T.L., 2006, Investigating the depth of komatiite melting using experimentally determined olivine and orthopyroxene melt reaction coefficients: American Geophysical Union, Fall Meeting, 2006, abstract V24B-05.
Bickle, M.J., Nisbet, E.G., and Martin, A., 1994, Archean greenstone belts are not oceanic crust: Journal of Geology, v. 102, p. 121-138.
Blichert-Toft, J., and Arndt, N.T., 1999, Hf isotope compositions of komatiites: Earth Planetary Science Letters, v. 171, p. 439-451.
Bouquain, S., Arndt, N.T., Libourel, G., and Foley, S., 2006, High-Ca augites do not prove that komatiites were wet: American Geophysical Union, Fall Meeting, 2006, abstract V24B-04.
Brandon, A.D., Walker, R.J., Puchtel, I.S., Becker, H., Humayun, M., and Revillon, S., 2003, 186Os-187Os systematics of Gorgona Island komatiites: Implications for early growth of the inner core: Earth Planetary Science Letters, v. 206, p. 411-426.
Campbell, I.H., Griffiths, R.W., and Hill, R.I., 1989, Melting in an Archean mantle plume: Heads it is basalts and tails it is komatiites: Nature, v. 339, p. 697-699.
Condie, K.C., 2001, Mantle Plumes and their Record in Earth history: Cambridge University Press, Cambridge, 306 p.
Dann, J.C., 2001, Vesicular komatiites, 3.5-Ga Komati Formation, Barberton Greenstone Belt, South Africa: Inflation of submarine lavas and origin of spinifex zones: Bulletin of Volcanology, v. 63, p. 462-481.
Dann, J.C., and Grove, T.L., 2007, Volcanology of the Barberton greenstone belt, South Africa: Inflation and evolution of flow field, in van Kranendonk, M.J., Smithies, R.H. and Bennett, V.C., eds., Precambrian Ophiolites and Related Rocks: Developments in Precambrian Geology, v. 15, Elsevier, Amsterdam, p. 527-570.
de Wit, M.J., and Ashwal, L.D., 1997, Preface: Convergence towards divergent models of greenstone belts, in de Wit, M. J. and Ashwal, L. D., eds., Greenstone Belts: Oxford University Press, Oxford, p. 9-17.
Dixon, J.E., Leist, L., Langmuir, C., and Schilling, J.G., 2002, Recycled dehydrated lithosphere observed in plumeinfluenced mid-ocean-ridge basalt: Nature, v. 420, p. 385-389.
Donaldson, C.H., 1982, Spinifex-textured komatiites: A review of textures, mineral compositions, and layering, in Arndt, N.T., and Nisbet, E.G., eds., Komatiites: George Allen and Unwin, London, p. 211-244.
Dostal, J., and Mueller, W.U., 1997, Komatiitic flooding of a rifted Archean rhyolitic arc complex: Geochemical signatures and tectonic significance of the Stoughton-Roquemaure Group, Abitibi Greenstone belt: Journal of Geology, v. 105, p. 545-563.
Dostal, J. and Mueller, W.U., 2004, Komatiite geochemistry, in Eriksson, P.G., Altermann, W., Nelson, D.R., Mueller, W.U. and Catuneanu, O., eds., The Precambrian Earth: Tempos and Events: Developments in Precambrian Geology, v. 12, Elsevier, Amsterdam, p. 290-298.
Fan, J., and Kerrich, R., 1997, Geochemical characteristics of aluminum depleted and undepleted komatiites and HREE-enriched low-Ti tholeiites, western Abitibi greenstone belt: A heterogeneous mantle plume-convergent margin environment: Geochimica et Cosmochimica Acta, v. 61, p. 47234744.
Faure, F., Arndt, N. and Libourel, G., 2006, Formation of spinifex texture in komatiites: An experimental study: Journal of Petrology, v. 47, p. 5911610.
Glotov, A.L., Polyakov, G.V., Hoa, T.T., Balykin, P.A., Akimtsev, V.A., Krivenko, A.P., Tolstykh, N.D., Phuong, N.T., Thanh, H.H., Hung, T.Q., and Petrova, T.E., 2001, The Ban Phuc Ni-Cu-PGE deposit related to the Phanerozoic komatiite - basalt association in the Song Da rift, northwestern Vietnam: Canadian Mineralogist, v. 39, p. 573-589.
Green, D.H., Nicholls, I.A., Viljoen, M., and Viljoen R., 1975, Experimental demonstration of the existence of peridotitic liquids in earliest Archean magmatism: Geology, v. 3, p. 11-14.
Grove, T.L., and Parman, S.W., 2004, Thermal evolution of the Earth as recorded by komatiites: Earth Planetary Science Letters, v. 219, p.173-187.
Grove, T.L., de Wit, M.J., and Dann, J.C., 1997, Komatiites from the Komati type section, Barberton, South Africa, in de Wit, M. J. and Ashwal, L. D., eds., Greenstone Belts: Oxford University Press, Oxford, p. 438-453.
Hanski, E., Walker, R.J., Huhma, H., Polyakov, G.V., Balykin, P.A., Hoa, T.T., Phuong, N.T., 2004, Origin of the Permian-Triassic komatiites, northwestern Vietnam: Contributions to Mineralogy and Petrology, v. 147, p. 453-469.
Herzberg, C.T., 1992, Depth and degree of melting of komatiites: Journal of Geophysical Research, v. 97, p. 45214540.
Herzberg, C.T., 1995, Generation of plume magmas through time: An experimental perspective: Chemical Geology, v. 126, p. 1-16.
Hill, R.E.T., Barnes, S.J., Gole, M.J., and Dowling, S.E., 1995, The volcanology of komatiites as deduced from field relationships in the Norseman-Wiluna greenstone belt, Western Australia: Lithos, v. 34, p.159-188.
Hofmann, A., 1998, Chemical differentiation of the Earth: The relationship between mantle, continental crust and oceanic crust: Earth Planetary Science Letters, v. 90, p. 297-314.
Inoue, T., Rapp, R.P., Zhang, J., Gasparik, T., Wedner, D.J., and Irifune, T., 2000, Garnet fractionation in a hydrous magma ocean and the origin of Aldepleted komatiites: Melting experiments of hydrous pyrolites with REEs at high pressure: Earth Planetary Science Letters, v. 177, p. 81-87.
Jochum, K.P., Arndt, N.T., and Hofmann, A.W., 1991, Nb-Th-La in komatiites and basalts: Constraints on komatiite petrogenesis and mantle evolution: Earth Planetary Science Letters, v. 107, p. 272-289.
Kerr, A.C., 2005, La Isla de Gorgona, Columbia: A petrological enigma?: Lithos, v. 84, p. 77-101.
Kerr, A.C., and Arndt, N.T., 2001, A note on the IUGS reclassification of the high-Mg and picritic volcanic rocks: Journal of Petrology, v. 42, p. 21692171.
Kerr, A.C., Marriner, G.F., Arndt, N.T., Tarney, J., Nivia, A., Saunders, A.D., and Duncan, R.A., 1996, The petrogenesis of Gorgona komatiites, picrites, and basalts: New field, petrographic and geochemical constraints: Lithos, v. 37, p. 246-260.
Lahaye, Y., and Arndt, T.N., 1996, Alteration of a komatiite flow from Alexo, Ontario, Canada: Journal of Petrology, v. 37, p. 1261-1284.
Lahaye, Y., Arndt, N.T., Byerly, G., Chauvel, C., Fourcade, S., and Gruau, G., 1995, The influence of alteration on the trace element and Nd isotopic compositions of komatiites: Chemical Geology, v. 126, p. 43-64.
Le Bas, M.J., 2000, IUGS Reclassification of the high-Mg and picritic volcanic rocks: Journal of Petrology, v. 41, p. 1467-1470.
Lesher, C.M., and Arndt, N.T., 1995, REE and Nd isotope geochemistry, petrogenesis and volcanic evolution of contaminated komatiites at Kambalda, Western Australia: Lithos, v. 34, p. 127-157.
Naldrett, A. J., 2004, Magmatic Sulphide Deposits: Springer-Verlag, Berlin, 727 p.
Nesbitt, R.W., Sun, S.S., and Purvis, A.C., 1979, Komatiites: geochemistry and genesis: Canadian Mineralogist, v. 17, p. 165-186.
Nisbet, E.G., Cheadle, M.J., Arndt, N.T., and Bickle, M.J., 1993, Constraining the potential temperature of the Archean mantle: A review of evidence from komatiites: Lithos, v. 30, p. 291-307.
Parman, S.W., Dann, J.C., Grove, T.L., and de Wit, M.J., 1997, Emplacement conditions of komatiite magmas from the 3.49 Ga Komati Formation, Barberton greenstone belt, South Africa: Earth Planetary Science Letters, v. 150, p. 303-323.
Richter, F.M., 1988, A major change in the thermal state of the Earth at the Archean-Proterozoic boundary: Consequences for the nature and preservation of continental lithosphere: Journal of Petrology, Special Lithosphere Issue, p. 39-52.
Rollinson, H., 2007, Early Earth Systems: A Geochemical Approach: Blackwell Publishing, Massachusetts, USA, 285 p.
Shimizu, K., Komiya, T., Hirose, K., Shimizu, N., and Maruyama, S., 2001, Cr-spinel, an excellent micro-container for retaining primitive melts-implications for a hydrous plume origin for komatiites: Earth Planetary Science Letters, v. 189, p. 177-188.
Shimizu, K., Nakamura, E., Kobayashi, K., and Maruyama, S., 2004, Discovery of Archean continental and mantle fragments inferred from xenocrysts in komatiites, the Belingwe greenstone belt, Zimbabwe: Geology, v. 32, p. 285-288.
Shore, M., and Fowler, A.D., 1999, The origin of spinifex texture in komatiites: Nature, v. 397, p. 691-693.
Sproule, R.A., Lesher, C.M., Ayer, J.A., Thurston, P.C., and Herzberg, C.T., 2002, Spatial and temporal variations in the geochemistry of komatiites and komatiitic basalts in the Abitibi greenstone belt: Precambrian. Research, v. 115, p. 153-186.
Sun, S.S., and McDonough, W.F., 1989, Chemical and isotopic systematics of oceanic basalts: Implications for mantle composition and processes: Geological Society, London, Special Publications, v. 42, p. 313-345.
Sylvester, P.J., Harper, G.D., Byerly, G.R., and Thurston, P.C., 1997, Volcanic Aspects, in de Wit, M.J., and Ashwal, L. D., eds., Greenstone Belts: Oxford University Press, Oxford, p. 55-90.
Thurston, P.C., and Chivers, K.M., 1990, Secular variation in greenstone sequence development emphasizing Superior Province, Canada: Precambrian Research, v. 46, p. 21-58.
Viljoen, M.J., and Viljoen, R.P., 1969, The geology and geochemistry of the lower ultramafic unit of the Onverwacht Group and a proposed new class of igneous rocks: Geological Society of South Africa Special Publication, v. 2, p. 55-86.
Wilson, A.H., Shirey, S.B., and Carlson, R.W., 2003, Archean ultra-depleted komatiites formed by hydrous melting of cratonic mantle: Nature, v. 423, p. 858-861.
Xie, Q., and Kerrich, R., 1994, Silicate-perovskite and majorite signature komatiites from Archean Abitibi Belt: Implications for early mantle differentiation and stratification: Journal of Geophysical. Research, v. 99, p. 15,799-15,812.